General Chemistry I Practical Exam
General Chemistry I Practical Exam
Name:
CRN:
| Points earned | |
| Identification | |
| Measurement | |
| Lab Technique | |
| Data Analysis | |
| Total points |
1. Identification:
- Classify the following mixtures as homogeneous (solutions) or heterogeneous:
| Homogeneous or heterogeneous | 0 points | 5 point | |
| Mixture 1 | incorrect | correct | |
| Mixture 2 | incorrect | correct | |
| Mixture 3 | incorrect | correct | |
| Mixture 4 | incorrect | correct |
- Match the glassware to the label.
| 0 points | 4 points | 8 points | |
| 250 mL Erlenmeyer flask | Wrong equipment | Wrong size | Correct equipment and size |
| 50 mL beaker | Wrong equipment | Wrong size | Correct equipment and size |
| 10 mL graduated cylinder | Wrong equipment | Wrong size | Correct equipment and size |
| Weighing boat | Wrong equipment | Correct equipment | |
| Pipette | Wrong equipment | Correct equipment |
2. Measurement:
- A.Weigh out 2.5 g of NaCl. Record your final measurement: __________________
- Record the volume of a liquid in a 10 mL graduated cylinder: ________________
- Record the volume of a liquid in a 50 mL graduated cylinder: ________________
- Record the temperature of a liquid using a thermometer: ___________________
| 0 points | 2 points | 6 points | 10 points | |
| Weigh out 2.5 g of NaCl. | Unable to perform skill | Within ±0.50 g and not recorded | Within ±0.10 g and recorded with mistakes | Within ±0.10 g and recorded correctly |
| Record the volume of a liquid in a 10 mL graduated cylinder. | Wrong number | Wrong sensitivity and no units | Wrong sensitivity or no units | Correct |
| Record the volume of a liquid in a 50 mL graduated cylinder. | Wrong number | Wrong sensitivity and no units | Wrong sensitivity or no units | Correct |
| Record the temperature of a liquid using a thermometer. | Wrong number | Wrong sensitivity and no units | Wrong sensitivity or no units | Correct |
3. Basic lab technique with waste disposal:
- Select all the equipment for gravity filtration. Demonstrate how it works.
- Select all the equipment and materials required for determining if a substance is acidic, basic or neutral. Demonstrate how it works.
- Select all the equipment and materials required to make a saturated alum solution. Demonstrate how it is done.
- Select all the equipment and materials required to determine the heat exchanged using a constant pressure (coffee cup) calorimeter. Demonstrate how it is done.
- Propose a method for collecting the gas formed in a reaction. Select the equipment required for the process.
| 0 points | 2 points | 6 points | 10 points | |
| Gravity filtration | Unable to start | Major mistakes | Small mistakes | Correct |
| Classification of acids and bases | Unable to start | Major mistakes | Small mistakes | Correct |
| Saturated solution | Unable to start | Major mistakes | Small mistakes | Correct |
| Coffee-cup calorimeter | Unable to start | Major mistakes | Small mistakes | Correct |
| Gas collection | Unable to start | Major mistakes | Small mistakes | Correct |
General Chemistry I Practical Exam
(Written portion)
Name:
CRN:

4. Data Analysis:
- Create a 5-layer density column using any of the substances listed in the table.
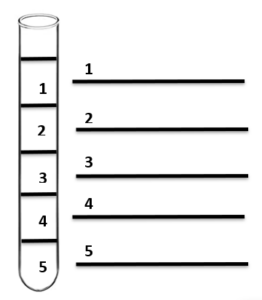
| Substance | Density
(g/mL) |
| Water | 1.00 |
| Dichloromethane | 1.33 |
| Vegetable oil | 0.92 |
| Honey | 1.42 |
| Dish soap | 1.06 |
| Baby oil | 0.83 |
| Pancake syrup | 1.37 |
| Corn syrup | 1.33 |
| Milk | 1.03 |
| Grain alcohol | 0.81 |
| Blood | 1.6 |
- A student separated a mixture of salt and sand to recover 16.32 g of salt and 7.44 g of sand. Calculate the percent composition of the mixture.
- A student performs the decomposition of baking soda starting with 137.53 g of baking soda (NaHCO3) and upon the completion of the experiment collects 82.11 g of sodium carbonate. Calculate the percent yield of the reaction using the balanced chemical reaction below.
NaHCO3(s) ![]() Na2CO3(s) + CO2(g) + H2O(g)
Na2CO3(s) + CO2(g) + H2O(g)
- A student reacts 5.63 g of baking soda (NaHCO3) with 25 mL of 0.83 M acetic acid solution. Identify the limiting reactant in this experiment.
- Fill out the following table by proposing an experiment that will help determine if a substance is polar or nonpolar. Using your previous experience working with that substance, classify the substance as polar or nonpolar.
| Substance | Method for determination | Polar or nonpolar? |
| Sugar |
| 0 points | 2 points | 6 points | 10 points | |
| Density | Unable to start | Major mistakes | Small mistakes | Correct |
| Percent composition | Unable to start | Major mistakes | Small mistakes | Correct |
| Percent yield | Unable to start | Major mistakes | Small mistakes | Correct |
| Limiting reactant | Unable to start | Major mistakes | Small mistakes | Correct |
| Polarity | Unable to start | Major mistakes | Small mistakes | Correct |